Professor of Cooperative Extension
Outreach web site: ucbiotech.org clear-project.org
Ph.D. University of Michigan, 1977
B.S. Miami University, 1968
Professor Peggy Lemaux’s laboratory performs both basic and applied research focused primarily on cereal crops, like sorghum, wheat, rice and barley. The objectives of these studies are to better understand crop plants and to use that knowledge to improve their performance and quality. More recently efforts with colleagues have focused on bioenergy and climate change – especially in the versatile feedstock, sorghum.
 The Lemaux laboratory focuses on development and use of genetic and genomic technologies for monocotyledonous species, like sorghum (Sorghum bicolor),, wheat (Triticum aestivum), barley (Hordeum vulgare) and the model C4 grass, Setaria viridis. Long-term objectives are to use these technologies to study gene functionin order to explore basic biological questions and to improve crops.
The Lemaux laboratory focuses on development and use of genetic and genomic technologies for monocotyledonous species, like sorghum (Sorghum bicolor),, wheat (Triticum aestivum), barley (Hordeum vulgare) and the model C4 grass, Setaria viridis. Long-term objectives are to use these technologies to study gene functionin order to explore basic biological questions and to improve crops.
Methods for stably transforming cereal crops are more routine today than decades ago, but challenges still exist. Nearly all methods utilize in vitro- tissue culture materials, which leads directly or indirectly to limitations in varieties that can be transformed, to somaclonal variation and to transgene expression instability.
Most cereal transformation efforts involve culturing immature embryos. This approach has been successful for model genotypes but, until recently, has been challenging for other varieties. For this reason, our laboratory developed other culturing methods, like cultured adventitious meristems and highly regenerable, green tissue, a developmental stage between embryogenic and organogenic tissue. These methods provide successful for previously recalcitrant varieties of wheat, barley, corn, rice, oat, sorghum and forage and turf grasses (e.g., Cho et al., 1998, 1999a; Ha et al., 2001; Cho et al., 2000a; Cho et al., 2000b; Zhang et al., 1999, 2002b; Gurel et al., 2009).
 One approach to study gene function was developed through an NSF-funded project in barley. The maize transposable element, Ds, and its transposase gene (with the late P. Bregitzer, USDA-ARS), were introduced into the same cell. Ds was activated and transposed preferentially to genic regions (Koprek et al., 2001; Brown et al., 2014). Inverse PCR was used to sequence flanking DNA, identify genes into which Ds inserted and map its location (Cooper et al., 2004; Singh et al., 2006). Efforts have continued to identify tagged genes in barley (Brown et al., 2014). Some of this work was continued by former postdoctoral fellow, Dr. Jaswinder Singh, now a professor at McGill University in Montreal.
One approach to study gene function was developed through an NSF-funded project in barley. The maize transposable element, Ds, and its transposase gene (with the late P. Bregitzer, USDA-ARS), were introduced into the same cell. Ds was activated and transposed preferentially to genic regions (Koprek et al., 2001; Brown et al., 2014). Inverse PCR was used to sequence flanking DNA, identify genes into which Ds inserted and map its location (Cooper et al., 2004; Singh et al., 2006). Efforts have continued to identify tagged genes in barley (Brown et al., 2014). Some of this work was continued by former postdoctoral fellow, Dr. Jaswinder Singh, now a professor at McGill University in Montreal.
Considerable effort was expended to create transgenic cereals that over- and under-expressed elements of the NADPH thioredoxin (Trx) system (with B. Buchanan, UCB). To achieve maximal Trx overexpression in the grain, we used seed-specific promoters and vacuolar targeting to direct transgene expression to protein bodies of the endosperm (Cho et al., 1999b; Cho, M.-J., Lemaux P.G., Buchanan B.B. Production of proteins in plant seeds. Official Gazette of the United States Patent and Trademark Office Patents. Patent Number: US 07157629, January 2, 2007. The Regents of the University of California). This technology , termed ExpressTec, is currently being used by a Colorado-based company to produce clinical therapeutics.
A number of transgenic lines over- or under-expressing Trx were created. Homozygous Trx h5 transgenic barley seeds germinated faster, alpha-amylase levels rose earlier and levels of the starch-degrading enzyme, pullulanase, were higher (Cho et al., 1999) - important traits during the malting process. In wheat, homozygous lines overexpressing Trx h5 had lowered allergenicity in the gliadin fraction, assessed using a canine model (Li et al., 2009), and improved dough quality with poor quality wheat flour (unpublished). Unpublished work overexpressing Trx h5 in sorghum led to improved digestibility (Wong et al., unpublished), important because sorghum flour is the least digestible of all cereals. None of these products made it to the commercial market due to costs of reguatory approval, unattainable for academic scientists.
 Building on this with Chinese collaborators, using antisense to reduce trx h9 levels, preharvest sprouting was prevented (Li et al, 2009), causing costly losses in the U..S. and China each year. Work in Arabidopsis demonstrated that trxh9, unlike other thioredoxins, is located in cell membranes, possibly func tioning in cell-to-cell communication of redox state between endosperm, embryo and aleurone (Meng et al. 2010).
Building on this with Chinese collaborators, using antisense to reduce trx h9 levels, preharvest sprouting was prevented (Li et al, 2009), causing costly losses in the U..S. and China each year. Work in Arabidopsis demonstrated that trxh9, unlike other thioredoxins, is located in cell membranes, possibly func tioning in cell-to-cell communication of redox state between endosperm, embryo and aleurone (Meng et al. 2010).
 In 2015 the EPICON project, Epigenetic Control of Drought Response in Sorghum, began, funded by DOE BER. This collaborative project, between UC Berkeley, UCANR, the Joint Genome Institute and the Pacific Northwest National Laboratory, used transcriptomics, metabolomics, and proteomics to develop an in-depth understanding of drought tolerance in Sorghum bicolor in the field. In addition to assessing the impact on the plant during drought, this project has also focused on the involvement of the microbial community during drought. The first published study (with D. Coleman-Derr) was a high-resolution exploration of the longitudinal development of plant microbiomes during control and drought conditions (Yu et al. 2018). A drought-induced enrichment of monoderms, like Actinobacteria, occurs and metatranscriptomics showed that drought results in increases in transcription of specific metabolite transporters. This correlated with increased production of the same metabolites. in vivo colonization experiments demonstrated that inoculation with drought-enriched Actinomycetes leads to increased plant fitness, following drought stress.
In 2015 the EPICON project, Epigenetic Control of Drought Response in Sorghum, began, funded by DOE BER. This collaborative project, between UC Berkeley, UCANR, the Joint Genome Institute and the Pacific Northwest National Laboratory, used transcriptomics, metabolomics, and proteomics to develop an in-depth understanding of drought tolerance in Sorghum bicolor in the field. In addition to assessing the impact on the plant during drought, this project has also focused on the involvement of the microbial community during drought. The first published study (with D. Coleman-Derr) was a high-resolution exploration of the longitudinal development of plant microbiomes during control and drought conditions (Yu et al. 2018). A drought-induced enrichment of monoderms, like Actinobacteria, occurs and metatranscriptomics showed that drought results in increases in transcription of specific metabolite transporters. This correlated with increased production of the same metabolites. in vivo colonization experiments demonstrated that inoculation with drought-enriched Actinomycetes leads to increased plant fitness, following drought stress. 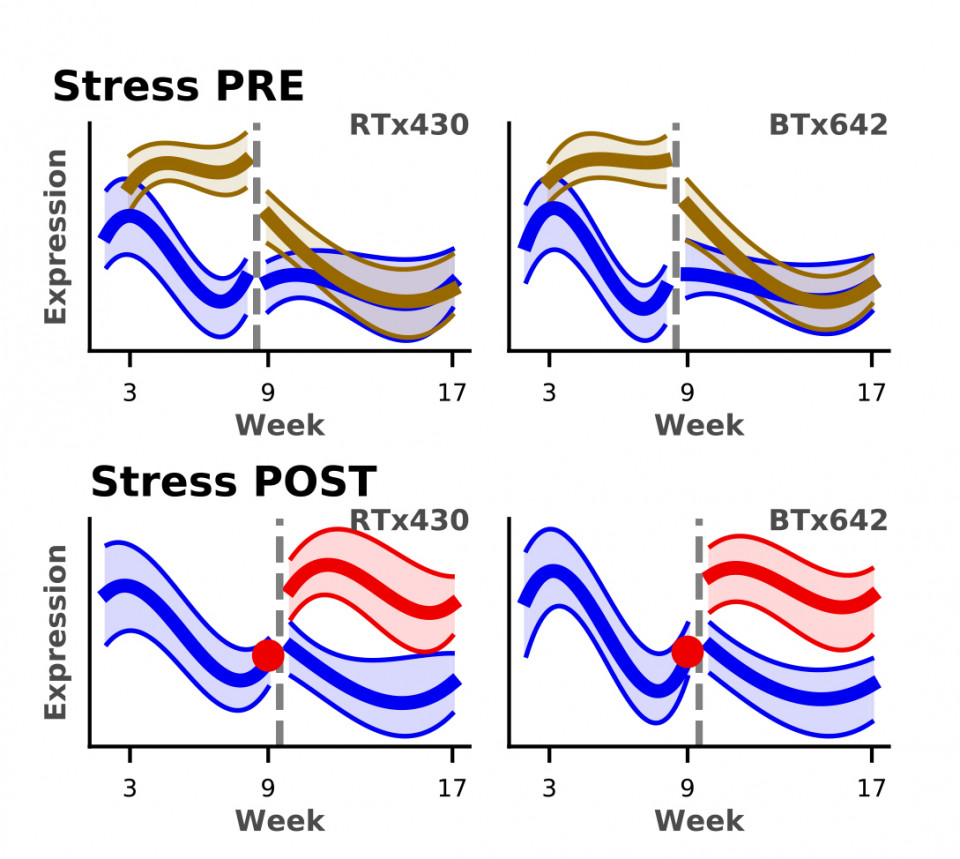 Companion studies (with J. Taylor) investigated the impact of drought on arbuscular mycorrhizal fungi, those microbes that provide plants with needed nutrients and mineral. These studies focused on describing fungal succession in nature and that drought causes changes in the dynamics of what fungi colonize the leaves and roots (Gao et al., 2019, 2020). Transcriptomics studies of leaf and root samples over the entire developmental phase and drought period revealed a large impact on transcription with >40% of gene expression being influenced by pre- and post-flowering drought (Varoquaux et al. 2019). These changes affect genes involved in critical functions, like biotic and abiotic stress responses and photosynthesis. Specifically, we identified genotypic differences in core photosynthesis and reactive oxygen-species-scavenging pathways, possibly related to drought tolerance. Also noted was a large-scale depletion in the expression of genes critical to arbuscular mycorrhizal (AM) symbiosis. Currently all three years of transcriptomic data are being compiled to identify the critical genes involved in drought tolerance ( (with B. Cole, J. Vogel at JGI, E. Purdom, UCB Statistics). Work continues to focus on validating function of a myriad of genes identified as targets critical to drought tolerance.
Companion studies (with J. Taylor) investigated the impact of drought on arbuscular mycorrhizal fungi, those microbes that provide plants with needed nutrients and mineral. These studies focused on describing fungal succession in nature and that drought causes changes in the dynamics of what fungi colonize the leaves and roots (Gao et al., 2019, 2020). Transcriptomics studies of leaf and root samples over the entire developmental phase and drought period revealed a large impact on transcription with >40% of gene expression being influenced by pre- and post-flowering drought (Varoquaux et al. 2019). These changes affect genes involved in critical functions, like biotic and abiotic stress responses and photosynthesis. Specifically, we identified genotypic differences in core photosynthesis and reactive oxygen-species-scavenging pathways, possibly related to drought tolerance. Also noted was a large-scale depletion in the expression of genes critical to arbuscular mycorrhizal (AM) symbiosis. Currently all three years of transcriptomic data are being compiled to identify the critical genes involved in drought tolerance ( (with B. Cole, J. Vogel at JGI, E. Purdom, UCB Statistics). Work continues to focus on validating function of a myriad of genes identified as targets critical to drought tolerance.
Additional information on EPICON is presented below.
Drought
Duration and intensity of drought is increasing in California and worldwide largely due to climate change. Faculty member Peggy Lemaux led a $12.3 million Department of Energy Biological and Environmental Research-funded project to examine how the drought-tolerant cereal crop, Sorghum bicolor, survives water limitation.
Lemaux and fellow PMB researchers John Taylor, Devin Coleman-Derr and John Vogel, partnered with Elizabeth Purdom, UCB Department of Statistics, Jeff Dahlberg and Robert Hutmacher, directors of ANR Research and Extension Centers, Axel Visel from DOE’s Joint Genome Institute and Christer Jansson, former Director of Plant Sciences at DOE’s Pacific Northwest National Laboratory.
EPICON
 “Epigenetic Control of Drought Response in Sorghum" (EPICON) started following an historic drought in California. During the four years of field-testing, leaf and root samples were taken weekly from germination to harvest under three watering conditions – control, pre-flowering and post-flowering drought. These samples were used to investigate responses of sorghum and its microbiome to drought, including transciptomic, metabolomic, proteomic, metagenomic and metatranscriptomic changes. While originally launched to look at epigenetic changes, that allow plants to respond to changing environments quickly, the richness of the transcriptomic and microbiome data became a more tractable focus,.
“Epigenetic Control of Drought Response in Sorghum" (EPICON) started following an historic drought in California. During the four years of field-testing, leaf and root samples were taken weekly from germination to harvest under three watering conditions – control, pre-flowering and post-flowering drought. These samples were used to investigate responses of sorghum and its microbiome to drought, including transciptomic, metabolomic, proteomic, metagenomic and metatranscriptomic changes. While originally launched to look at epigenetic changes, that allow plants to respond to changing environments quickly, the richness of the transcriptomic and microbiome data became a more tractable focus,.
It has long been known that associations of specific bacteria and fungi with plants have positive effects on plant fitness. Through this detailed temporal and spatial assessment of the microbial communities in leaves, roots, rhizosphere and soil, we were able to develop a more informed picture of the role of microbes in the drought responses of plants, and of the influence of the communication channels between these groups of organisms.

"Recent efforts that grew out of information from the EPICON studies are focused on developing better predictions about how sorghum will be affected by future climate scenarios, and how using the reslience of sorghum to these climate chalenges can inform efforts to better equip other crops to address climate change. These efforts, currently funded by outside organizations including the Chan-Zuckerberg Initiative, are aimed at developing improved means to sequester carbon in the soil through improved photosynthetic efficiency leading to even deeper roots and improved ways to sequester that added carbon in the soil through manipulation of plant-mirobial interactions.
Outreach Interests
 As a Professor of Cooperative Extension, I have statewide responsibility for outreach and educational programming related to agriculture and food. These efforts are designed to increase public understanding of agricultural practices, food production and the impact of new technologies, including biotechnology and genome editing, on food and agriculture. Educational programming and resources that are developed include development of an award winning, website, http://ucbiotech.org, which provides science-based information and resources. Three articles related to these efforts were published in Annual Review of Plant Biology, “Genetically Engineered Crops and Foods: A Scientist's Analysis of the Issues. Part I and II (Lemaux PG. 2008; 2009). and "Genetic Engineering and Editing of Plants: An Analysis of New and Persisting Questions" (Mackelprang and Lemaux 2020) were published in Annual Reviews of Plant Biology. With my long-time assistant, B. Alonso, we created games, displays, videos and middle-school afterschool curricula, DNA for Dinner, Backyard Mystery and Learning Plant Biology: It’s All in the Touch. The latter became increasingly popular during the pandemic, when parents were looking for easy-to-use lessons to do with their children. The CLEAR (Communication, Literacy, Education for Agricultural Research) project was initiated in late 2015 with funds from UCOP’s Global Food Initiative. CLEAR aims to enable undergrads, grads and postdocs with skills in communicating about science with the media, legislators and the public. CLEAR members write pieces for the popular press, organize roundtables, participate in outreach events, develop videos and convene science presentations at local bars in their PubScience (https://clear-project.org/pubscience/) efforts.
As a Professor of Cooperative Extension, I have statewide responsibility for outreach and educational programming related to agriculture and food. These efforts are designed to increase public understanding of agricultural practices, food production and the impact of new technologies, including biotechnology and genome editing, on food and agriculture. Educational programming and resources that are developed include development of an award winning, website, http://ucbiotech.org, which provides science-based information and resources. Three articles related to these efforts were published in Annual Review of Plant Biology, “Genetically Engineered Crops and Foods: A Scientist's Analysis of the Issues. Part I and II (Lemaux PG. 2008; 2009). and "Genetic Engineering and Editing of Plants: An Analysis of New and Persisting Questions" (Mackelprang and Lemaux 2020) were published in Annual Reviews of Plant Biology. With my long-time assistant, B. Alonso, we created games, displays, videos and middle-school afterschool curricula, DNA for Dinner, Backyard Mystery and Learning Plant Biology: It’s All in the Touch. The latter became increasingly popular during the pandemic, when parents were looking for easy-to-use lessons to do with their children. The CLEAR (Communication, Literacy, Education for Agricultural Research) project was initiated in late 2015 with funds from UCOP’s Global Food Initiative. CLEAR aims to enable undergrads, grads and postdocs with skills in communicating about science with the media, legislators and the public. CLEAR members write pieces for the popular press, organize roundtables, participate in outreach events, develop videos and convene science presentations at local bars in their PubScience (https://clear-project.org/pubscience/) efforts.
Research:
Aregawi, K., Shen, J., Pierroz, G., Bucheli, C., Sharma, M., Dahlberg, J., Owiti, J., Lemaux, P.G. 2021. Morphogene-assisted Transformation of Sorghum bicolor Allows More Efficient Genome Editing. Plant Biotech J 20 (4): 748-760 https://doi.org/10.1111/pbi.13754
Scavuzzo-Duggan, T., Varoquaux, N., Ortega, J., Belcher, M., Madera, M., Vogel, J., Dahlberg, J., Hutmacher, R., Coleman-Derr, D., Lemaux, P.G., Purdom, E., Scheller, H.F. 2021. Cell wall compositions of Sorghum bicolor leaves and roots remain relatively constant under drought conditions. Front Plant Sci Nov12 https://doi.org/10.3389/fpls.2021.747225
Xu, L., Dong, Z., Chiniquy, D., Pierroz, G., Deng, S., Gao, C., Diamond, S., Simmons, T., Wipf, H M-L., Caddell, D., Varoquaux, N., Madera, M.A., Hutmacher, R., Deutschbauer, A., Dahlberg, J.A., Guerinot, M.L., Purdom, E., Banfield, J.F., Taylor, J.W., Lemaux, P.G., Coleman-Derr,,D..2021. Genome resolved metagenomics reveals role of iron metabolism in drought-induced rhizosphere microbiome dynamics. Nature Comm. https://doi.org/10.1038/s41467-021-23553-7
Xu, L., Pierroz, G., Wipf, H. M-L, Gao, C. Taylor, J.W., Lemaux, P.G., Coleman-Derr, D. 2021. Holo-omics for deciphering plant-microbiome interactions. Microbiome 9:69 https://doi.org/10.1186/s40168-021-01014-z
Wipf, H., Xi, L., Gao, C., Spinner, H., Taylor, J., Lemaux, P.G., Mitchell, J., Coleman-Derr, D. 2020. Agricultural Soil Management Practices Differentially Shape the Bacterial and Fungal Microbiome of Sorghum bicolor. App Env Micro Dec 11:87(5):e02345-20. https://doi.org/10.1128/AEM.02345-20
Outreach:
Lemaux P.G. 2013. “The What & How of Classic and Modern Methods of Plant Modification”. The Soy Connection 21: 4.
Lemaux P.G. 2013.”Plant Biology Researchers Face Their Own Fiscal Cliffs” ASPB News 40 (1): 1-3,8.
Lemaux P.G. 2013. “Egg-citing! ASPB’s STEPs at the White House” ASPB News 40 (3):1-4.
Lemaux P.G. 2013. “To Die For?” ASPB News 40 (5): 1-4.
Lemaux, P.G. 2010. What's Slowing Commercialisation of GE Crops? Regulatory, Economic, Intellectual Property and Consumer Acceptance Issues. In '1st Australian Summer Grains Conference'. Gold Coast, Australia. (Eds B George-Jaeggli, DJ Jordan). (Grains Research and Development Corporation).
Charles Reid Barnes Lifetime Achievement Award, ASPB, 2022.
Charles Reid Barnes Lifetime Achievement Aware, ASPB 2022
Award of Excellence, Extension Education Community Education Materials, ASA-CSSA-SSA, November 2015
American Society of Plant Biologists Excellence in Education Award, 2012
President, American Society of Plant Biologists, 2011
Society for In Vitro Biology 2010 Lifetime Achievement Award - Society for In Vitro Biology - 2010
Fellow - Crop Science Society of America - 2007
Distinguished Service Award, Outstanding Outreach - Cooperative Extension Academic Assembly Council, Division of Agriculture and Natural Resources - 2006
Dennis R. Hoagland Award for outstanding contribution to agricultural research - American Society of Plant Biologists - 2003
Fellow - American Association for the Advancement of Science - 2002
Distinguished Service Award, Outstanding Research - Cooperative Extension Academic Assembly Council, Division of Agriculture and Natural Resources - 1997
Honored Women of the University of California, Berkeley - UC Berkeley - 1995
Peggy G. Lemaux
Berkeley, California 94720