Professor Emeritus
The Fischer Lab studies imprinted genes expressed from their maternal alleles in the endosperm. MET1 DNA methyltransferase methylates and silences imprinted genes. DEMETER antagonizes MET1, allowing maternal alleles to be expressed. DEMETER is a DNA glycosylase and is expressed specifically in the central cell, a maternal gamete fertilized to form the endosperm. We study how DEMETER antagonizes MET1, what restricts DEMETER expression to the central cell, how DEMETER targets imprinted genes, and the identity and function of new imprinted genes.
Honors and Awards
Fellow - American Society of Plant Biology - 2018
Distinguished Teaching Award - College of Natural Resources - 2010
Member - National Academy of Sciences - 2009
Fellow - American Association for the Advancement of Science - 2007
Service
2011 - 2014 Chair Department of Plant & Microbial Biology
2010 - 2011 Interim Chair Department of Plant & Microbial Biology
1989 - 1990 Chair Division of Plant Biology
Regulation of Plant Gene Imprinting
The primary goal of the research in my laboratory is to understand how DNA demethylation regulates gene imprinting. DNA methyltransferases methylate DNA (5-methylcytosine), which silences transposons, repeated sequences, and genes. In Arabidopsis, the base excision DNA repair pathway is used to actively demethylate DNA. The DEMETER (DME) DNA glycosylase initiates the pathway by removing 5-methylcytosine through cleavage of the N-glycosylic bond. AP endonuclease, DNA polymerase, and ligase insert cytosine and seal the DNA, resulting in a loss of DNA methylation.
Gene imprinting is an important process for reproduction in plants. Alleles of imprinted genes are expressed differently depending on whether they are inherited from the male or female parent. In mammals, imprinted genes contribute to the control of fetal growth and development, and human diseases are linked to mutations in imprinted genes. In plants, imprinted genes control the growth and development of seeds, which are the primary source of carbon, nitrogen, and energy for humans and domesticated animals.
In both plants and mammals DNA methylation (5-methylcytosine) is a critical regulator of gene imprinting. In mammals, allele-specific silencing by de novo DNA methylation establishes gene imprinting in the embryo. In plants, allele-specific activation by DNA demethylation establishes DNA imprinting in the endosperm. Because endosperm does not contribute to the next generation, the activated allele need not be silenced again. Double fertilization enables plants to use such “one-way” control of imprinting and DNA methylation in endosperm.
In Arabidopsis, DME specifically excises 5-methylcytosine at the MEDEA (MEA) Polycomb group gene and activates its transcription in the central cell of the female gametophyte. After fertilization of the central cell, the maternal expression of the MEA Polycomb group protein then maintains the silenced state of its own paternal allele and additional imprinted genes in the endosperm.
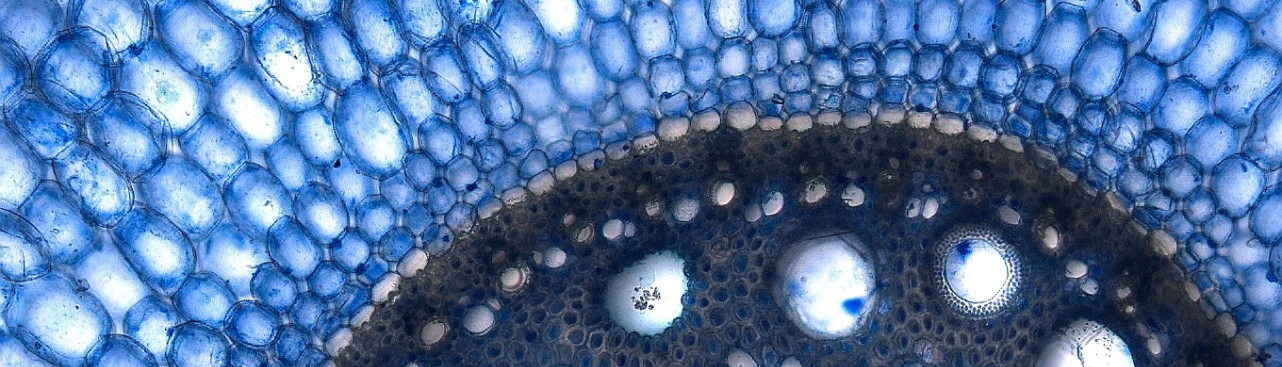
Green fluorescing central cells within red Arabidopsis ovules. The DEMETER DNA glycosylase establishes
gene imprinting in the central cell by demethylating the MEA Polycomb maternal allele.
DME related proteins ROS1 (REPRESSOR OF SILENCING1), DML2 and DML3 (DEMETER-LIKE2 and 3) are broadly expressed in vegetative and reproductive tissues. They actively demethylate approximately 200 sites in the Arabidopsis genome. Their function may be to protect genes from being silenced by preventing the spread of DNA methylation from neighboring transposons and repeated sequences. This process may prevent the accumulation of excessive DNA methylation over successive generations that could interfere with transcription of genes.
Parent-of-origin-specific (imprinted) gene expression is regulated in Arabidopsis thaliana endosperm by cytosine demethylation of the maternal genome mediated by the DNA glycosylase DEMETER, but the extent of the methylation changes is not known. We found that virtually the entire endosperm genome is demethylated, coupled with extensive local non-CG hypermethylation of small interfering RNA–targeted sequences. Mutation of DEMETER partially restores endosperm CG methylation to levels found in other tissues, indicating that CG demethylation is specific to maternal sequences. Endosperm demethylation is accompanied by CHH hypermethylation of embryo transposable elements. Our findings demonstrate extensive reconfiguration of the endosperm methylation landscape that likely reinforces transposon silencing in the embryo.
We used extensive sequencing of cDNA libraries to identify 9 paternally expressed and 34 maternally expressed imprinted genes in A. thaliana endosperm that are regulated by the DNA-demethylating glycosylase DEMETER, the DNA methyltransferase MET1, and/or the core Polycomb group protein FIE. These genes encode transcription factors, proteins involved in hormone signaling, components of the ubiquitin protein degradation pathway, regulators of histone and DNA methylation, and small RNA pathway proteins. We also identified maternally expressed genes that may be regulated by unknown mechanisms or deposited from maternal tissues. We did not detect any imprinted genes in the embryo. Our results show that imprinted gene expression is an extensive mechanistically complex phenomenon that likely affects multiple aspects of seed development.
Recent Publications
Zhang, C., Hung, Y.-H., Rim, H.J., Zhang, D., Frost, J.M., Shin, H., Jang, H., Liu, F., Xiao, W., Iyer, L.M., Aravind, L., Zhang, X.-Q, Fischer, R.L., Huh, J.H., and Hsieh, T.-F. (2019) The catalytic core of DEMETER guides active DNA demethylation in Arabidopsis. Proc. Natl. Acad. Sci. USA. in press.
Kim, Y.M., Ono, A., Kinoshita, T., Zilberman, D., Scholten, S., Okamoto, T., Fischer, R.L. (2019) DNA demethylation by ROS1a in rice vegetative cells promotes methylation in sperm. Proc. Natl. Acad. Sci. USA. Published online, doi:10.1073/pnas.1821435116.
Frost, J.M., Kim, Y.M., Park, G.-T., Hsieh, P.-H., Nakamura, M., Lin, S., Yoo, H., Choi, J., Ikeda, Y., Kinoshita, T., Choi, Y., Zilberman, D., Fischer R.L. (2018) The FACT complex is required for DNA demethylation at heterochromatin during reproduction in Arabidopsis. 2018 Proc. Natl. Acad. Sci. USA. Published online, May 15;115(20):E4720-E4729. doi: 10.1073/pnas.1713333115.
Park, J-.P., Frost, J.M., Park, K., Ohr, H., Park, G.T., Kim, S., Eom, H., Lee, I., Brooks, J.S., Fischer, R.L. and Choi, Y. (2017) Control of DEMETER DNA demethylase gene transcription in male and female gamete companion cells in Arabidopsis thaliana. Proc. Natl. Acad. Sci. USA. 114:2078-2083.
Hsieh, P.-H., He., S., Buttress, T., Gao, H., Couchman, M., Fischer, R.L., Zilberman, D., Feng, X. (2016) The Arabidopsis male sexual lineage exhibits more robust maintenance of CG methylation than somatic tissues. Proc. Natl. Acad. Sci. USA. 113:15132-15137.
Park, K., Kim, M.Y., Vickers, M., Park. J.-S., Hyun, Y., Okamoto, T., Zilberman, D., Fischer, R.L., Feng, X., Choi, Y., Scholten, S. (2016) DNA demethylation is initiated in the central cells of Arabidopsis and rice. Proc. Natl. Acad. Sci. USA. 113:15138-15143.
Mérai, Z., Chumak, N., García-Aguilar, M., Hsieh, T.-H., Nishimura, T., Schoft, V.K., Bindics, J., Ślusarz, L., Arnoux, S., Opravil, S., Mechtler, K., Zilberman, D., Fischer, R.L. and Tamaru H. (2014) Cdc48/p97 disassembles centromeres, decondenses heterochromatin and activates ribosomal RNA genes. Proc. Natl. Acad. Sci. USA. 111:16166-17171.
Brooks, S., Huh, J., Fischer, R.L., Eichman, B. (2014) 5-methylcytosine recognition by Arabidopsis thaliana DNA glycosylases DEMETER and DML3. Biochemistry 53:2525-2532.
Rodrigues, J.A., Ruan, R., Nishimura, T., Sharma, M., Sharma, R., Ronald, P.C., Fischer, R.L., Zilberman, D. (2013) Imprinted expression of genes and small RNA is associated with localized hypomethylation of the maternal genome in rice endosperm. Proc. Natl. Acad. Sci. USA 110:7934-7939.
Ibarra, C.A., Feng, X., Schoft, V.K., Hsieh, T.-F., Uzawa, R., Rodrigues, J.A., Zemach, A., Chumak, F., Machlicova, A., Nishimura, T., Rojas, D., Fischer, R.L., Tamaru, H., Zilberman, D. (2012) Active DNA demethylation in plant companion cells reinforces transposon methylation in gametes. Science 337:1360-1364.
Schoft, V.K., Chumak, N., Choi, Y., Hannon, M., Garcia-Aguilar, M., Machlicova, A., Slusarz, L. Mosiolek, M., Park, J.-S., Park, G.T., Fischer, R.L., Tamaru, H. (2011) Function of the DEMETER DNA Glycosylase in the Arabidopsis thaliana Male Gametophyte. Proc. Natl. Acad. Sci. USA 108:8042-8047.
Bauer, M. and Fischer, R.L. (2011) Genome Demethylation and Imprinting in the Endosperm. Current Opinion in Plant Biology. 14:1-6.
Hsieh, T.-F., Shin, J., Uzawa, R., Silva, P., Cohen, S., Bauer, M.J., Hashimoto, M., Kirkbride, R., Harada, J.J., Zilberman, D., Fischer, R.L. (2011) Regulation of Imprinted Gene Expression in Arabidopsis Endosperm. Proc. Natl. Acad. Sci. USA 108:1755-1762.
Mok, Y.G., Uzawa, R., Lee, J., Weiner, G.M., Eichman, B.F., Fischer, R.L., Huh, J.H. (2010) Domain structure of the DEMETER 5-methylcytosine DNA glycosylase. Proc. Natl. Acad. Sci. USA 107:19225-19230.
Hsieh, T.-F., Ibarra, C.A., Silva, P., Zemach, A., Eshed-Williams, L., Fischer, R.L., Zilberman, D. (2009) Genome-wide Demethylation of Arabidopsis Endosperm. Science. 324:1451-1454.
Huh, J.H., Bauer, M.J., Hsieh, T.F., and Fischer, R.L. (2008) Cellular Programming of Plant Gene Imprinting. Cell. 132:735-744.
Penterman, J., Zilberman, D., Huh, J.H., Ballinger, T., Henikoff, S., and Fischer, R.L. (2007) DNA Demethylation in the Arabidopsis Genome Proc. Natl. Acad. Sci. USA. 104:6752-6757.
Huh, J.H., Bauer, M.J., Hsieh, T.-F., Fischer, R.L. (2007) Endosperm Gene Imprinting and Seed Development. Current Opinion Genetics and Development. 17:480-485.
Xiao, W., Custard, K.D., Brown, R.C., Lemmon, B.E., Harada, J.J., Goldberg, R.B., Fischer, R.L. (2006) DNA Methylation Is Critical for Arabidopsis Embryogenesis and Seed Viability. Plant Cell 18:805-814.
Gehring, M., Huh, J. H., Hsieh, T.-F., Penterman, J., Choi, Y., Harada, J.J., Goldberg, R.B., Fischer, R.L. (2006) DEMETER DNA glycosylase establishes MEDEA Polycomb gene self-imprinting by allele-specific demethylation. Cell 124:495-506.
Xiao, W., Brown, R.C., Lemmon, B.E., Harada, J.J., Goldberg, R.B., Fischer, R.L. (2006) Regulation of Seed Size by Hypomethylation of Maternal and Paternal Genomes. Plant Physiology. 142:1160-1168.
Hsieh, T.-F. and Fischer, R.F. (2005) Biology of Chromatin Dynamics. Annual Review of Plant Biology. 56:327-351.
Choi, Y., Harada, J.J., Goldberg, R.B., Fischer, R.L. (2004) An invariant aspartic acid in the DNA glycosylase domain of DEMETER is necessary for transcriptional activation of the imprinted MEDEA gene. Proc. Natl. Acad. Sci. USA. 101:7481-7486.
Kinoshita, T., Miura, A., Choi, Y., Kinoshita, Y., Cao, X., Jacobsen, S.E., Fischer, R.L., and Kakutani, T. (2004) One-way control of FWA imprinting in Arabidopsis endosperm by DNA methylation. Science. 303:521-523.
Gehring, M., Choi, Y., and Fischer, R.L. (2004) Imprinting and Seed Development. Plant Cell. 16 Supplement:S203-S213.
Xiao, W., Gehring, M., Choi, Y., Margossian, L., Pu, H., Harada, J.J., Goldberg, R.B. Pennell, R.I., and Fischer, R.L. (2003) Imprinting of the MEA Polycomb Gene Is Controlled by Antagonism Between MET1 Methyltransferase and DME Glycosylase. Developmental Cell. 5:891-901.
Reiser, L., Fischer, R.L. (1993) The ovule and embryo sac. Plant Cell. 5:1291-1301.
Choi, Y., Gehring, M., Johnson, L., Hannon, M.., Harada, J.J., Goldberg, R.B., Jacobsen, S.E., Fischer, R.L. (2002) DEMETER, a DNA Glycosylase Domain Protein, is Required for Endosperm Gene Imprinting and Seed Viability in Arabidopsis. Cell. 110:33-42.
Kinoshita, T., Harada, J.J., Goldberg, R.B., Fischer, R.L. (2001) Polycomb Repression of Flowering during Early Plant Development. Proc. Natl. Acad. Sci. USA 98:14156-14161.
Mizukami, Y. and Fischer, R.L. (2000) Plant organ size control: AINTEGUMENTA regulates growth and cell numbers during organogenesis. Proc. Natl. Acad. Sci. USA. 97: 942-947.
Kinoshita, T., Yadegari, R., Harada, J.J., Goldberg, R.B. and Fischer, R.L. (1999) Imprinting of the MEDEA polycomb gene in the Arabidopsis endosperm. Plant Cell 11:1945-1952.
Ohad, N., Yadegari, R., Margossian, L., Hannon, M., Michaeli, D., Harada, J.J., Goldberg, R.B., Fischer, R.L. (1999) Mutations in FIE, a WD Polycomb Group Gene, Allow Endosperm Development Without Fertilization. Plant Cell 11:407-415.
Kiyosue, T., Ohad, N., Yadegari, R., Hannon, M., Dinneny, J., Wells, D., Katz, A., Margossian, L., Harada, J.J., Goldberg, R.B., Fischer, R.L.F. (1999) Control of fertilization-independent endosperm development by the MEDEA Polycomb gene in Arabidopsis. Proc. Natl. Acad. Sci. USA 96:4186-4191.
Lotan, T., Ohto, M.-A., Yee, K.M., West, M.A.L., Lo, R., Kwong, R.W., Yamagishi, K., Fischer, R.L., Goldberg, R.B., Harada, J.J. (1998) Arabidopsis LEAFY COTYLEDON1 is sufficient to induce embryo development in vegetative cells. Cell 93:1195-1205.
Ohad, N., Margossian, L., Hsu, Y.-C., Williams, C., Repetti, P., Fischer, R.L. (1996) A Mutation that Allows Endosperm Development Without Fertilization. Proc. Natl. Acad. Sci. USA 93: 5319-5324.
Klucher, K.M., Chow, H., Reiser, L., Fischer, R.L. (1996) The AINTEGUMENTA Gene in Arabidopsis Required for Ovule and Female Gametophyte Development is Related to the Floral Homeotic Gene APETALA2. Plant Cell 8: 137-153.
Reiser, L., Modrusan, Z., Samach, A., Margossian, L., Ohad, N., Haughn, G., Fischer, R.L. (1995) The BELL1 gene encodes a homeodomain protein involved in pattern formation in the Arabidopsis ovule primordium. Cell 83:735-742.
Modrusan, Z., Reiser, L., Feldmann, K.A., Fischer, R.L., Haughn, G.W. (1994) Homeotic transformation of ovules into carpel-like structures in Arabidopsis. Plant Cell 6:333-349.
Robert Fischer
Berkeley, California 94720